Research
Publications
Group Pictures
Conferences
Family Album
David L. Tierney, Associate Professor
Miami University (2009-present)
University of New Mexico (2001-2009)
NIH Postdoctoral Fellow,
Northwestern University (1996-2000)
Ph.D., University of Michigan (1996)
B.S., Saginaw Valley State University (1989)
Office: 165A Hughes Laboratories
Phone: (513) 529-8234
e-mail: tiernedl@miamioh.edu

We have three interrelated research areas:
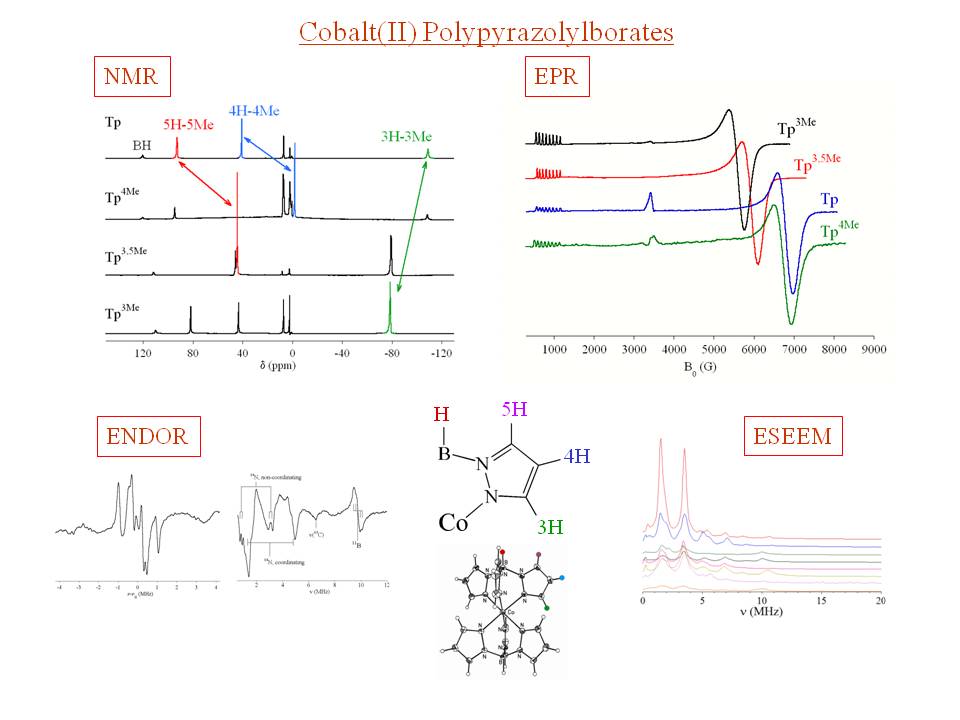
Our main focus is in the bioinorganic chemistry of zinc, geared to the elucidation of structure/function relationships in metalloenzyme active sites to gain a deeper understanding of enzyme mechanism. Our overriding interest is in the use of Co(II) as a spectroscopic probe of metal sites in proteins. The Co(II) ion is particularly well-suited to the study of Zn biochemistry, as substitution of Co(II) for Zn(II) usually returns an enzyme that is fully functional. Unlike the colorless, non-magnetic 3d10 Zn(II) ion, Co(II) (3d7) is paramagnetic in all environments and it's complexes are highly colored. Favorable electronic properties make Co(II) complexes accessible by nearly all magnetic resonance techniques.
However, the extraction of structural properties from spectroscopic observations is often hindered by the complicated electronic structure of high-spin Co(II), including the presence of substantial
spin-orbit coupling in the ground state and thermally accessible excited states.
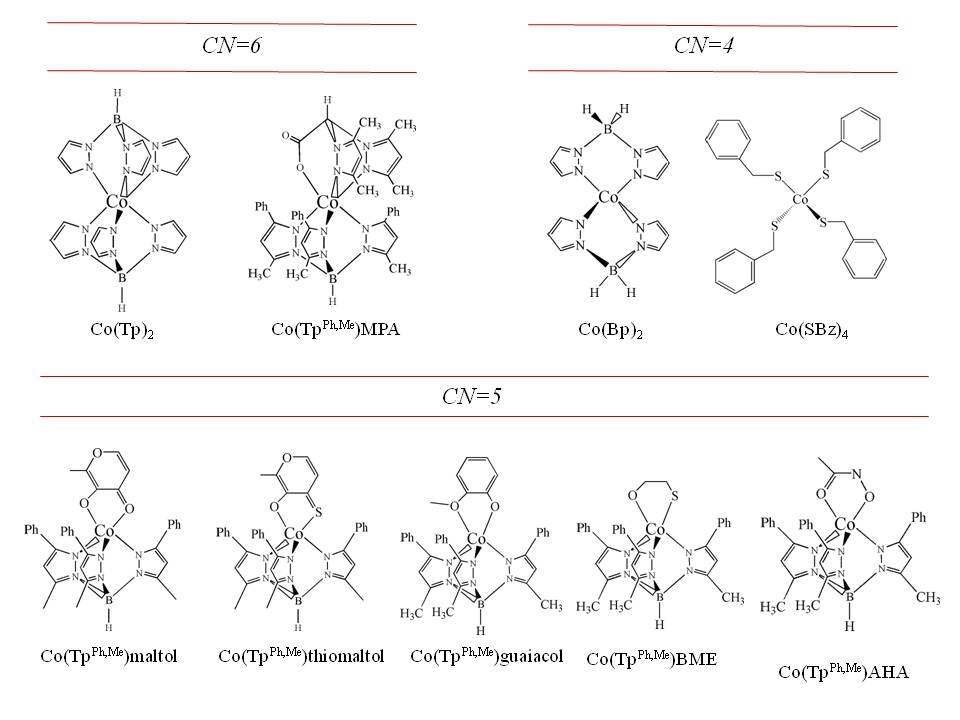
To better understand the correlation between structure and spectroscopy, we are involved the preparation of an ever-growing library of Co(II) model compounds, encompassing four-, five- and six-coordination, with varying contributions from N, O and S donors (examples below). Development of these structural correlations involves characterization of both the physical and electronic structure of
the metal site. We use X-ray crystallography to determine the three-dimensional structure of a complex, and UV-visible-near infrared spectroscopy to determine the ground state electronic structure. This information is necessary to understand the magnetic resonance properties of the molecule.
Ours is one of the only labs in the world that integrates all variations of paramagnetic resonance:
(1) Synthesis and spectroscopy of high-spin Fe(II) and Co(II) coordination complexes.
(2) Paramagnetic resonance spectroscopy of Co(II)-substituted Zn(II) proteins.
(3) X-ray absorption spectroscopy of Zn(II) proteins and their Co(II)-substituted analogs.
electron paramagnetic resonance (EPR), at both X-band (9 GHz) and Q-band (35 GHz)
electron-nuclear double resonance (ENDOR), both continuous wave and pulsed, at both X-band and Q-band
electron-spin-echo envelope modulation (ESEEM), at both X-band and Q-band
nuclear magnetic resonance (NMR), variable temperature at 200, 300, 500 and 850 MHz
NMR relaxometry (a. k. a. NMR dispersion, NMRD) on a home-built low-field, high resolution spectrometer
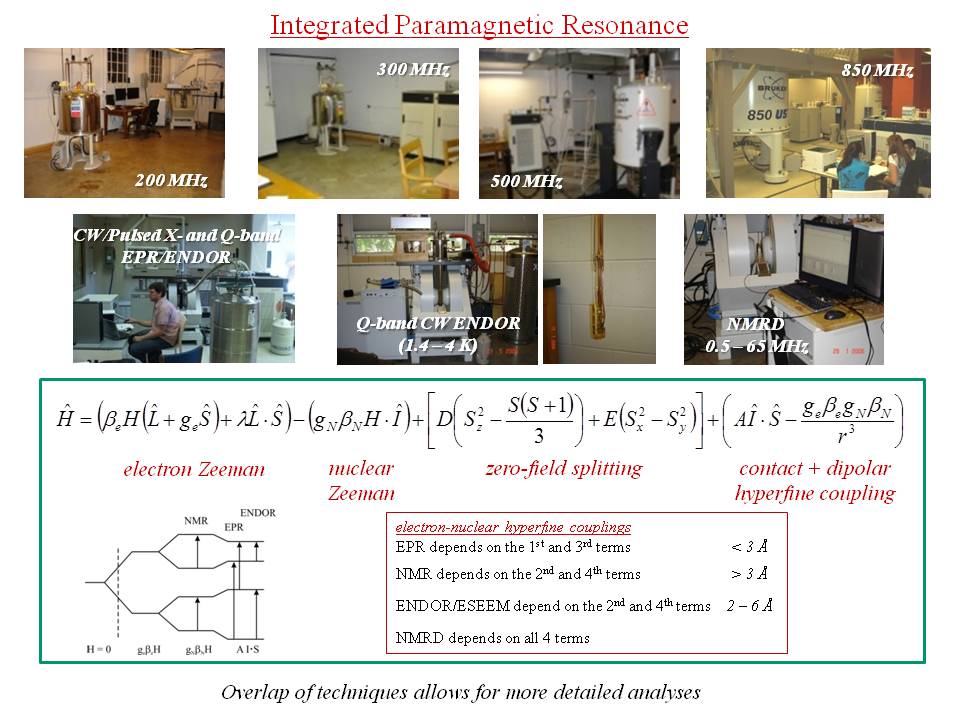
All of the above is available in-house, either in our lab, or in the
Ohio Advanced EPR Lab and the
High Field Magnetic Resonance Lab. The concurrent application of all of these techniques affords an unprecedented level of structural and bonding detail through interrogation of electron-nuclear hyperfine couplings from both points of view - that of the electron, and that
of the nucleus. Each approach gives unique information; each has its limitations. For example, EPR and ENDOR, which give information
regarding the symmetry and ligation of a metal ion, are typically performed at cryogenic temperatures on
frozen solutions, offering access to trapped intermediates. In contrast, NMR and NMRD experiments, which give information regarding
more distant structural features, are carried out at or near room temperature on fluid solutions, offering access to dynamic information.
We use the suite of techniques to study the metal sites in Zn enzymes, in two main areas. The newest of these is in the development of targeted inhibitors for matrix metalloproteinases (MMPs), which have been implicated in the metastasis of cancers and other tumors, through MMP-mediated breakdown of connective tissues. Through the study of small molecule binding to the related enzyme carbonic anhydrase, which similarly binds Zn(II) in a tris-histidine, monoaquo geometry, and its Co(II)-substituted form, we are identifying zinc-binding groups that are more effective than the current clinical standard, acetohydroxamic acid (AHA), which we have shown to be fluxional. UV-visible studies determine the strength of binding, while magnetic resonance spectroscopies define the nature (mono- or bidentate, stable or fluxional) of metal-binding.
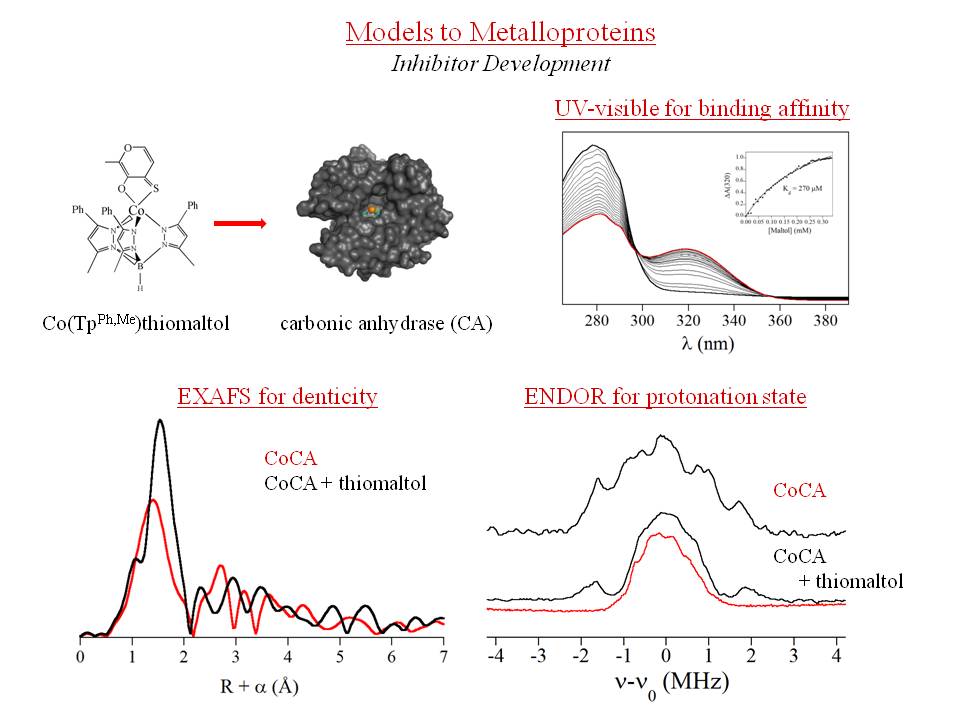
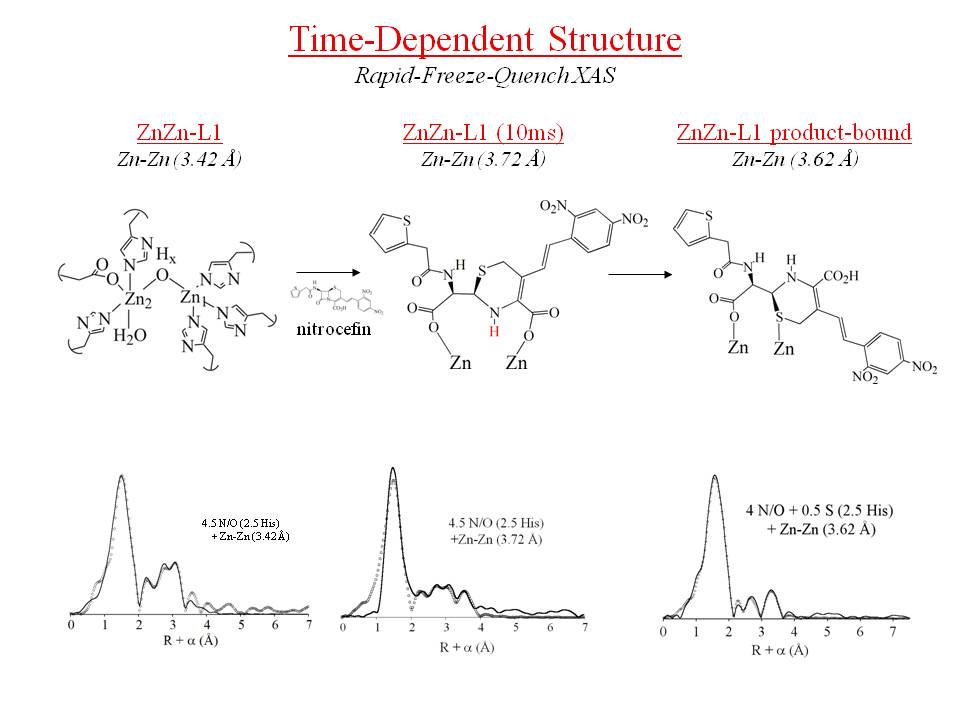
In addition, in collaboration with Prof. Crowder's lab, we are examining the chemistry of metallo-b-lactamases, which confer resistance to penicillins. All of these enzymes use one or two Zn ions at their active sites, and all of them remain active when Co is substituted for Zn. This project relies very heavily on x-ray absorption spectroscopy (XAS) to compare the structures of the native Zn and Co-substituted proteins, requiring us to travel to the National Synchrotron Light Source three times a year. Coupled with rapid-freezing techniques, we are using spectroscopy to take structural snapshots of the metal sites as a function of reaction coordinate, literally spying on enzymatic turnover at the atomic level. These studies are designed to define the key structural details that will lead to the development of effective mechanism-based inhibitors for these clinically important enzymes.